Scroll to:
Feline chlamydiosis (review)
https://doi.org/10.29326/2304-196X-2025-14-4-326-336
Abstract
Introduction. Feline chlamydiosis is a disease caused by Chlamydia felis, it is characterized by conjunctivitis (unilateral or bilateral), lacrimation and lesions of the respiratory and reproductive tracts. This pathogen is a gram-negative bacterium with a strictly obligate intracellular parasitic nature. In the early 1940s, Chlamydia were considered an intermediate link between viruses and bacteria. Until the 1960s, Chlamydia were classified as viruses due to their small size and inability to grow on artificial nutrient media. Their unique two-phase developmental cycle allows the pathogen to persist in the body of an animal or human for a long time without clinical manifestations. Treatment of chlamydiosis must be systemic, addressing both etiological therapy (use of antibiotics) and symptomatic therapy. Immunity against chlamydiosis is weak, with cellular immunity being more important than humoral immunity. Currently, both attenuated and inactivated vaccines are available on the market, which can protect against the clinical manifestation of the disease but not against infection. Chlamydiosis is a globally widespread disease, with detection rates showing a consistent year-on-year increase.
Objective. To review and systematize current data on feline chlamydiosis caused by Chlamydia felis.
Results. The article reviews global prevalence data of feline chlamydiosis. It examines the biological properties of the infectious agent, the clinical signs of the disease, and the pathological findings. Data on immunity are presented, and disease control measures are discussed.
Conclusion. Chlamydia felis is a worldwide spread pathogen capable of infecting not only cats and other animals but also humans, which indicates its zoonotic potential. Such factors as complex life cycle, adeptness at host immune evasion and ability to establish persistent infections hinder its effective eradication. The required extended treatment regimens and propensity for chronic infections compromise companion animal welfare and pose a risk of transmission to humans. A more profound understanding of Chlamydia felis pathogenesis is essential for developing effective treatment and prevention strategies.
For citations:
Tsyganov I.S., Shcherbinin S.V., Galkina T.S., Gruzdev K.N. Feline chlamydiosis (review). Veterinary Science Today. 2025;14(4):326-336. https://doi.org/10.29326/2304-196X-2025-14-4-326-336
INTRODUCTION
Feline chlamydiosis is a contagious disease of felids, characterized by ocular and respiratory tract lesions and caused by bacteria belonging to the genus Chlamydia. While the disease can be caused by different Chlamydia species, such as C. pneumoniae, С. psittaci, C. abortus, the primary agent is Chlamydia felis (previously known as Chlamydophila felis and earlier classified as Chlamydia psittaci variant felis). Given the historical classification of C. felis and C. psittaci as the same species, it can be assumed that past cases in domestic cats attributed to C. psittaci were indistinguishable from those caused by C. felis due to the limitations of molecular diagnostics at the time. Although C. psittaci and C. felis exhibit different host specificities and cause distinct clinical syndromes, these pathogens are closely related [1][2][3][4][5]. The reference strain for C. felis is the FP Baker strain [6][7][8][9][10][11]. This species is also a zoonotic pathogen capable of causing infection in humans [12][13][14]. Regarding host range, C. felis can asymptomatically infect dogs, with the manifestation of conjunctivitis being a rare event; however, seroconversion to this pathogen does occur [13]. Consequently, dogs may play a potential role in the zoonotic transmission of Chlamydia, making C. felis an important pathogen to diagnose in dogs as well. In humans, C. felis can cause symptoms of keratoconjunctivitis, follicular conjunctivitis, respiratory tract pathology, hepatosplenomegaly, glomerulonephritis and endocarditis [15][16][17][18][19][20]. It has also been reported that C. felis was detected in conjunctival swabs from an adult Eurasian lynx with unilateral ocular lesions [21].
In the early 1940s, Chlamydia were considered an intermediate link between viruses and bacteria. Until the 1960s, they were classified as viruses due to their small size and inability to grow on artificial nutrient media. At the beginning of the 21st century, after genome sequencing, this pathogen was classified as a bacterium [22][23]. The first reports of Chlamydia infection in cats with symptoms of ocular and respiratory system lesions were in 1971 [17].
The relevance of this work lies in the fact that the review presents the biology of the pathogen C. felis and describes the features of the disease caused by this pathogen, which is widespread almost everywhere among stray and domestic cats (Felis catus). This is facilitated by inadequate pet care standards, uncontrolled breeding, and expanded stray population. The topic is critically important for developing strategies to enhance the health of companion and breeding animals – a significant concern given the high costs of veterinary care – and for mitigating the zoonotic risk to humans. As chlamydiosis is found on all continents, improving diagnostic and preventive measures is essential.
The novelty of this article lies in its analysis of current data on the epizootiology, clinical presentation, pathology, diagnosis, and vaccine prevention of feline chlamydiosis.
The objective of this review is to update and systematize data on feline chlamydiosis caused by C. felis.
EPIZOOTIC SITUATION
Information on feline chlamydiosis epizootic situation does not reflect the true prevalence of the disease. Data on the frequency of feline chlamydiosis cases caused by C. felis from various geographical regions are based on molecular test results. For instance, in the United States in 2014, the percentage of cats testing positive for C. felis was 24% [24]. In China, between November 2022 and October 2023, the proportion of PCR-positive samples from cats was 15.75% [25]. In European countries over the last 20 years, the leaders in C. felis detection rates have been the following countries: Slovakia (45.16%), Hungary (33.3%), Poland (25%), Italy (20%), Switzerland (16%) and Sweden (15.3%) [9][13][26][27]. In Russia, from 2018 to September 2019, the proportion of positive results for C. feliswas 11.2% [28]; specifically for Moscow in 2019,it was 7.2% [29]. According to some reports, bacteria of this species were detected in approximately 26.3% of stray cats in Japan [26]. These data, without considering sampling and diagnostic rates, demonstrate a high incidence of chlamydiosis in cats. Cases caused by C. felis are characterized as enzootic, and epizootiological studies conducted in various countries indicate the presence of this pathogen in 23% of cats suffering from conjunctivitis. Higher morbidity rates are observed in cats kept in groups, such as in animal shelters [2].
BIOLOGICAL PROPERTIES OF THE PATHOGEN
Chlamydia felis is a gram-negative, coccoid bacterium with strictly obligate intracellular parasitism. It belongs to the family Chlamydiaceae, genus Chlamydia (Fig.). Its size depends on the phase of the developmental cycle, which occurs in two stages: non-infectious reticulate bodies – RB (diameter from 0.5 to 1.6 µm) and infectious elementary bodies – EB (diameter from 0.2 to 0.6 µm) [7][30].
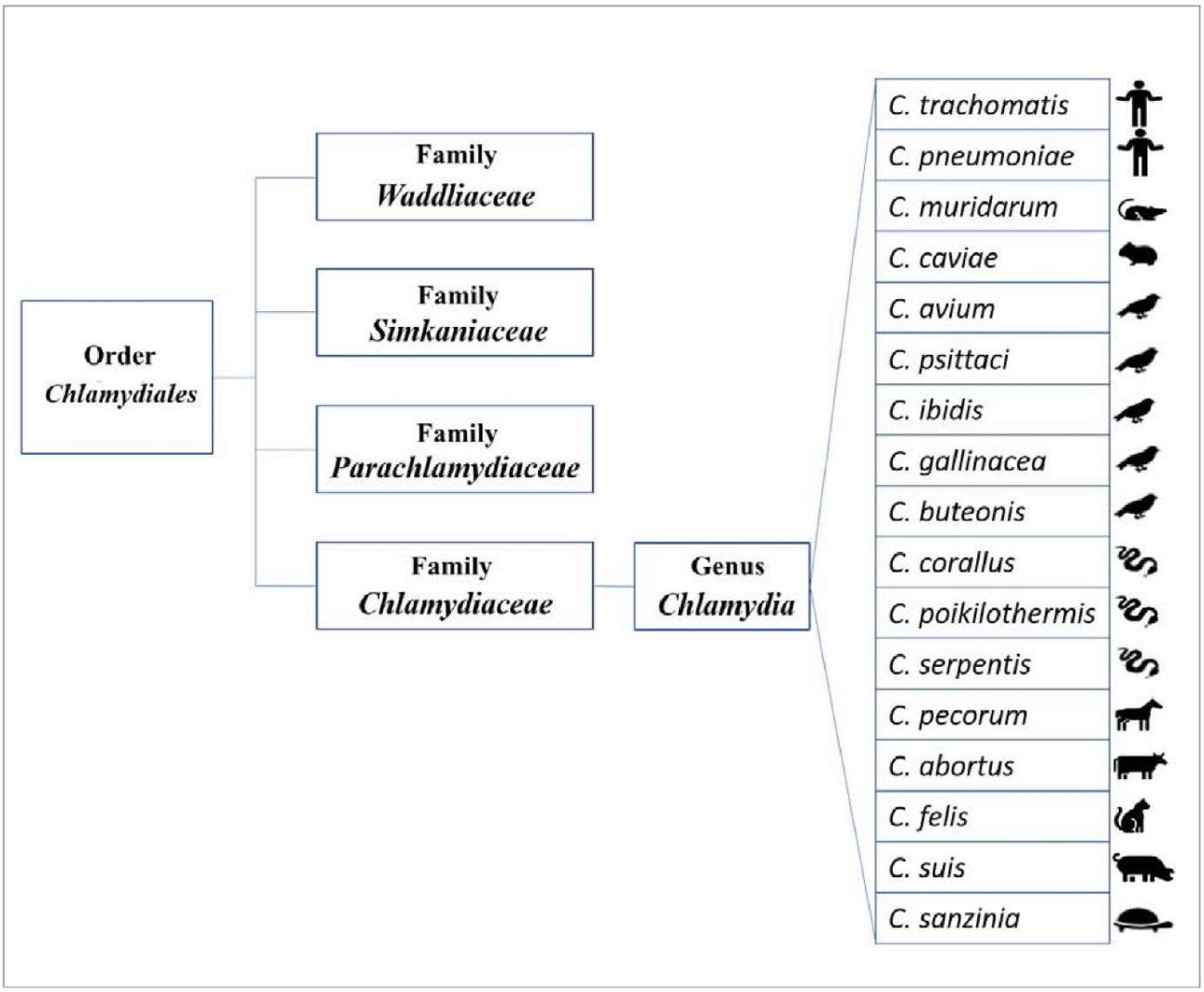
Fig. Phylogenetic classification of Chlamydia species according to 16S and 23S rRNA gene sequence analysis as of 2019 [32]. Icons indicate the primary host species for each strain, though all members of the Chlamydia genus are considered polyhostal
Initially, all chlamydial isolates obtained from cats with upper respiratory symptoms since 1942 were classified as C. psittaci. Subsequent 16S rRNA sequence analysis, however, led to a reclassification of the family Chlamydiaceae into two genera: Chlamydia and Chlamydophila, with the feline agent being placed in the latter [8][22][30][31]. This taxonomy was revised again in 2009 following whole-genome sequencing studies, which consolidated all pathogenic chlamydias into the single family Chlamydiaceae and a single genus, Chlamydia, within the expanded order Chlamydiales [11].
PATHOGEN RESISTANCE TO ENVIRONMENTAL FACTORS AND DISINFECTANTS
Chlamydias are inactivated by heat, with exposure to 55, 70, and 75 °C proving lethal after 45, 2, and 1 minute, respectively. While low temperatures have a preservative effect, the pathogen is susceptible to disinfectants (e.g., formaldehyde, chloramine, phenol) in standard concentrations that are effective against chlamydias [28][32]. Low pH levels are detrimental, with an optimal pH range of 7.0–7.4. Chlamydias can persist in tap water at room temperature for 2–3 days and are sensitive to ultraviolet radiation [33].
MOLECULAR BIOLOGICAL CHARACTERISTICS OF THE PATHOGEN
The causative agent of chlamydiosis has a single circular chromosome with a genome size of just over 1.166 million base pairs; the guanine-cytosine content is 39.1–39.4%. It also contains a cryptic plasmid, pCfe1, approximately 7.6 thousand base pairs in size, with eight genes [31][34].
The chlamydial genome is highly conserved and compact, a feature characteristic of C. felis as well [20][35]. Due to their intracellular lifestyle, chlamydias have undergone genome reduction, which is an adaptation rather than degradation [22]. The chlamydial envelope consists of three layers: an inner bilipid membrane, an intermembrane structure (periplasmic space), and an outer lipopolysaccharide membrane containing inclusions of the major outer membrane protein (MOMP) and polymorphic outer membrane proteins (POMP – pmp1, pmp7, pmp13, and others), as well as cysteine-rich proteins. POMP includes the outer membrane proteins OMP1 and OMP2. These proteins are involved in the pathogen’s adhesion to host epithelial cells [31][34][33][36]. MOMP and OMP2 proteins contain genus-, species-, and type-specific epitopes, which can lead to cross-reactions. The MOMP protein (also known as OmpA – outer membrane protein A) is found in both EB and RB and functions as both an adhesin and a porin [31][35][37]. Outer membrane proteins are highly conserved among isolates [38].
The chlamydial envelope lacks peptidoglycan but contains a polypeptide that, together with covalently bound lipoproteins, lipopolysaccharides, and outer membrane proteins, provides mechanical strength to the envelope [26][31][39]. Experimental data show that polymorphic membrane proteins (pmp) possess multiple biological and immune functions, including evasion of the host immune response, induction of inflammatory and immune reactions, and tissue tropism. The pmp genes show a high degree of intra- and interspecies heterogeneity in their amino acid sequences and sizes, but they can be identified by the presence of conserved amino acid sequences GGA(I, L, V) and FxxN, repeated in the N-terminal region and the C-terminal translocation block / β-domain [40][41][42]. The number of pmp genes varies among Chlamydia species, ranging from 9 to 21; 12 have been identified in C. felis. The similarity between the nucleotide sequences of pmp is high, with non-synonymous single nucleotide polymorphisms (SNPs) limited to one site in each of the pmp1, pmp9, and pmp20 genes. It can be assumed that the pmp genes of C. felis are highly conserved among strains from different geographical regions [9][37][41][42]. Pmp1 and pmp7 are likely immunodominant proteins [42].
Cryptic plasmid genes were found in cats with severe clinical symptoms of chlamydial infection. The cryptic plasmid is often associated with chlamydial virulence [18][43]. The plasmids contain non-coding RNA and 8 open reading frames (ORF1–8), with the functions of 5 of them being sufficiently studied. Analysis of the nucleotide sequences of the open reading frames showed that the proteins encoded by ORF1 (pgp7) and ORF2 (pgp8) are homologs of integrase and recombinase, respectively, and are responsible for regulating plasmid replication, while ORF3 (pgp1) is an analog of DnaB helicase and is involved in unwinding double-stranded DNA during replication. ORF4 encodes the pgp2 protein, whose function has not yet been determined. One of the main frames is ORF5, encoding the pgp3 protein, which may be a marker of chlamydial infections. ORF6 encodes the pgp4 protein, containing 101–102 amino acids, and ORF7 and ORF8 encode the pgp5 and pgp6 proteins, respectively, which may be involved in plasmid replication. The plasmids encode various proteins involved in the replication of the plasmid itself, but it is not established whether they affect communication with the host cell; it has only been demonstrated that the pgp3 protein is secreted in the cytosol of Chlamydia-infected cells. The role of plasmids is not yet fully understood, although it has been shown that pgp1 (ORF3), pgp2 (ORF4), pgp6 (ORF8), and pgp8 (ORF2) are essential genes for plasmid maintenance, while pgp3 (ORF5), pgp5 (ORF7), and pgp7 (ORF1) are less essential [43].
Chlamydiae possess a specialized type III secretion system (T3SS), which allows these bacteria to inject virulence factors directly into the cytosol of the target cell of a susceptible organism, leading to disruption of adaptive immune response mechanisms (hindering the fusion of the phagosome with the host cell lysosome) [32][33]. Type III secreted effectors include the translocated actin-recruiting phosphoprotein, necessary at the initial stage of infection, the family of inclusion membrane proteins (internal compartments) required for the development of this inclusion, and the effector chlamydial outer protein N (CopN), involved in the late disruption of type III secretion activity. Other key factors include proteins playing a central role in chlamydial development and gene regulation, such as histone-like proteins HctA and HctB; cysteine-rich proteins (OmpA and OmpB); proteins responsible for the permeability of the outer membrane to nutrients (OmpA and PorB), as well as the global gene regulatory factor Euo [11].
Comparison of the C. felis genome with other members of the genus Chlamydia revealed that 795 genes are common to them, while 47 genes are unique to C. felis. It was also found that orthologous genes show a similar divergent pattern, except for 14 genes that have accumulated the most mutations, suggesting that these C. felis genes may be involved in evolutionary adaptation [31]. There is diversity among C. felis strains, which differ in their virulence [5][8].
Chlamydiae are characterized by a unique developmental cycle consisting of two phases: extracellular and intracellular. In the extracellular phase, the pathogen is represented by EBs, which are infectious. In the intracellular phase, the pathogen exists as non-infectious RBs [8][26][32]. Elementary bodies, responsible for infecting target cells, are protected by a three-layer envelope, which, together with their spherical shape, provides resistance to physical and chemical factors in the extracellular environment. Reticulate bodies are also spherical but have a reticulate structure, making them polymorphic and possessing a thin envelope [14][17][32]. Briefly, the reproduction cycle consists of sequential stages: 1) adsorption onto host cell receptors; 2) penetration of EBs into the cell via endocytosis and residence in a phagosome; 3) suppression of phagosome-lysosome fusion; 4) differentiation of EBs into RBs; 5) multiplication of RBs by binary fission; 6) differentiation of RBs into EBs; 7) release of EBs from the cell via lysis [44].
The cycle initiates when a susceptible host cell encounters the pathogen, the EBs first adhere to sialic acid on the cell membrane and then penetrate into the cytoplasm. Once inside, they form intracytoplasmic inclusions near the host cell nucleus, Golgi apparatus and endoplasmic reticulum. The metabolic activity of EBs, constrained by a limited set of enzymes, is sustained by the host cell’s reserves. From these reserves, EBs obtain the necessary metabolites and adenosine triphosphate (ATP) for their replication. Inside the inclusions, EBs transform into RBs, which then replicate via binary fission. Following replication, RBs differentiate back into infectious EBs. These new EBs are released from the host cell either by lysis or extrusion, enabling the infection to spread. The entire cycle is completed within 48 to 72 hours [8][17][30][31][32][33][45].
Currently, another stage of the chlamydial developmental cycle is recognized: the persistence stage. This latent form, also known as non-infectious aberrant bodies (ABs), is induced under stressful conditions, such as exposure to β-lactam antibiotics (e.g., penicillins, cephalosporins, carbapenems). In this state, the bacteria do not transform into EBs and become a non-replicating intracellular form; ABs are characterized by the absence of MOMP and the overproduction of stress proteins, notably Chsp60 (chlamydial heat shock protein 60, 60 kDa heat shock proteins). This response allows the infection to persist, contributing to chronic inflammation. The latent form is reversible if the stressor is removed: ABs will revert to the RB form, begin the replication phase, and continue development, transforming into EBs [31][45]. Reticulate bodies differ from EBs in that they are dedicated to nutrient uptake and replication, a process that can lead to their asynchronous differentiation back into EBs. The latter can persist for long periods in the intercellular space without being phagocytized. There is a constant resilience between the host and the pathogen, and some clinical chlamydial infections can be asymptomatic for several months [17][31][33].
Chlamydiae exhibit epitheliotropism. Chlamydial replication in epithelial cells leads to the destruction of the epithelial layer with the formation of scars and adhesions. During prolonged persistence in epithelial cells, the pathogen can enter the bloodstream and hematogenously spread to parenchymal organs and lymphoid tissues. C. felis affects the eyes and upper respiratory tract (nose or throat) of cats [8][33]. The pathogen is most commonly found in multi-cat households, shelters, and catteries. The main carriers of the pathogen are diseased cats, however, dogs can also serve as a reservoir for C. felis. Transmission occurs mainly through contact with infected material, such as ocular discharge [46]. There is evidence that animals can become infected through sexual contact. In female cats, the pathogen localizes in the cervix, while in male cats, the testes are infected and the pathogen is shed during ejaculation [47]. It was later proven that C. felis can be isolated from rectal and vaginal swabs taken from cats with natural and experimental chlamydial conjunctivitis, but the possibility of sexual transmission is unclear; likely, the intestine and reproductive organs may be sites of persistent infection [8][35]. Experimental ocular infection of cats led to the isolation of Chlamydiae from the vagina and rectum in 50% and 40% of cases, respectively, demonstrating that C. felis is not limited to the conjunctival mucosa [35].
PATHOGENESIS AND CLINICAL SIGNS OF CHLAMYDIOSIS
The incubation period ranges from 5 to 15 days [32]. Infection caused by C. felis leads to the development of acute or chronic conjunctivitis in cats. The infection typically begins in one eye and may subsequently spread to the other, resulting in bilateral conjunctivitis. Other clinical manifestations include unilateral or bilateral epiphora, hyperemia of the nictitating membrane, blepharospasm, mucoid/mucous/serous/seropurulent discharge, inflammation and chemosis of the conjunctival membranes. These symptoms persist for 22–45 days. Meanwhile, a mild form of conjunctivitis can last for several months [9][35][46][48][49]. Clinical signs from the respiratory system are usually minimal [48][49]. Some cats may experience weight loss. Fever, decreased or loss of appetite, lethargy, sneezing, serous nasal discharge, and enlargement of the submandibular lymph nodes are also possible [8][32][35]. The condition of most cats remains satisfactory, but a small number of animals develop serious respiratory distress, severe wheezing, which, if left untreated, can lead to death from pulmonary edema or asphyxia due to poorly developed mouth breathing. In the case of systemic infection, which is rare, the pathogen spreads through the bloodstream to almost all organs, joints, the brain and spinal cord, which is highly likely to be fatal [8][17][32]. Most untreated cats develop chronic conjunctivitis, which is characterized by mild conjunctival hyperemia and scant ocular discharge, or they may develop a follicular form of the condition [32][35]. Ocular discharge resolves within 60 days, but the absence of clinical symptoms does not guarantee recovery; chlamydial infection can persist asymptomatically for several months [17][35]. Co-infection caused by a combination of C. felis with other pathogens (feline calicivirus, feline herpesvirus type 1, mycoplasmas) increases the severity of the disease and the duration of chlamydial shedding into the environment. Chlamydial infection can manifest as peritonitis [8][50]. In experimental infection of cats, chlamydiae were detected in conjunctival swabs for up to 8 months after infection, suggesting a long period of asymptomatic carriage. Aerosol and oral administration of the pathogen to cats led to the development of upper respiratory tract pathology and mild gastritis. An association between chlamydial infection and lameness in sick cats has also been identified, manifesting two weeks after the onset of conjunctivitis in 10 out of 19 infected animals, although this fact requires more detailed study [8][17][35]. Furthermore, experimental inoculation of the pathogen into the genital tract resulted in chronic salpingitis with subsequent spread to the oviduct [46]. Literature data are conflicting regarding reproductive disorders; it has been suggested that the pathogen is a cause of abortions or missed pregnancy in cats. Some authors have indirect evidence that C. felis can cause abortion, neonatal mortality, and infertility, but definite cause-and-effect relationships have not been established [8][10]. Infertility and abortions are the most serious consequences of the chronic form of chlamydiosis [40]. It is believed that some metabolites (isoleucine) may act as inhibitors of chlamydial growth and contribute to the latent course of chlamydial infection [32]. Although chlamydiosis can be asymptomatic and usually not fatal, the infected animal remains a carrier capable of transmitting the pathogen [51].
PATHOLOGICAL CHANGES IN CHLAMYDIOSIS OF CATS
Post-mortem examination reveals the following pathological changes: hyperemia and enlargement of the liver and spleen, lymph nodes; signs of pneumonia and pericarditis; hemorrhages on serous membranes and under the renal capsule; lesions of the digestive tract. The most characteristic changes are observed in lung tissues, mediastinal and bronchial lymph nodes [32][47]. At the macroscopic level, the following phenomena are visible: serous conjunctivitis, hyperplasia of splenic lymphoid nodules, catarrhal bronchitis, subtotal and total interstitial pneumonia and pneumosclerosis, hyperplasia and serous lymphadenitis of regional lymph nodes. Histological examination reveals the following morphological criteria of chlamydial infection at the microscopic level: interstitial pneumonia, pneumosclerosis, fibrinous-purulent pleuropneumonia, interstitial bronchopneumonia (thickening of interstitial alveolar septa due to infiltration of inflammatory cells and exudate in bronchial tissue), hyperplasia and serous lymphadenitis of mediastinal and bronchial lymph nodes, hyperplasia of lymphoid nodules of the spleen and intestinal mucosa, venous hyperemia of the liver and kidneys, fatty and granular dystrophy of the liver. There is also marked congestion of organs with diapedesis phenomena around capillaries and hyperplasia of the islet regions secreting insulin in the pancreas. Mild external clinical symptoms may be combined with serious degenerative, destructive changes in internal organs and tissues [17][41][52].
LABORATORY DIAGNOSIS OF CHLAMYDIOSIS
Laboratory diagnosis is based on the direct detection of the pathogen in the material and the determination of anti-chlamydial antibodies through serological reactions (enzyme-linked immunosorbent assay, complement fixation test, long-term complement fixation test). Serological reactions in the diagnosis of chlamydiosis are screening tests requiring further confirmation by other methods. There are laboratory methods for detecting RBs and EBs. These include cytological (this analysis does not require special equipment, but its sensitivity and specificity are quite low), immunological (direct and indirect immunofluorescence assay, enzyme immunoassay), cultural (considered the gold standard but labor-intensive and time-consuming, used in scientific practice), and molecular biological (polymerase chain reaction – the most sensitive, rapid, and reliable) methods [33][53][54]. Molecular analysis is the most reliable for making a definitive diagnosis of “feline chlamydiosis”, as the disease can have various clinical manifestations and occur in a subclinical form (bacterial carriage) [51].
Due to the inability of Chlamydia to grow on artificial nutrient media, 6–7-day-old specific pathogen-free chicken embryos are used for their cultivation, infected into the yolk sac. Embryo death within the first two days after infection is considered non-specific. The incubation period lasts up to 13 days, which is its main disadvantage. Cultivation in chicken embryos until 1965 was the only method for isolating and propagating Chlamydia; however, the subsequent use of cell cultures simplified this procedure [17][55]. To reduce cultivation time to 48–72 hours, sensitive cell cultures are used; in particular, for growing C. felis, cell lines such as Vero (a continuous cell line from the kidney of the African green monkey Chlorocebus aethiops), McCoy (a hybrid line of human synovial cells and mouse fibroblasts), CrFK (a continuous cell culture of feline kidney), BHK-21 (a cell line from the kidney of a newborn Syrian hamster), HeLa (human cervical cancer cells), L929 (a mouse fibroblast cell line) are used [17][42][50]. To increase the adsorption and penetration of Chlamydia into the cell, centrifugation or physicochemical methods to reduce the resistance of cell cultures are used. For example, polycations (DEAE-dextran – diethylaminoethyl-dextran) are used to treat the monolayer before infecting cell cultures; they neutralize the anionic surface of Chlamydia, creating conditions for contact. After contact, antimetabolites are added to the nutrient medium, which slow down the metabolism of the cells but do not affect Chlamydia, indirectly stimulating their reproduction. Most often, Chlamydia are cultured on cell cultures treated with cycloheximide or analogs (L-cysteine hydrochloride, hydrocortisone, colchicine) [56]. Conjunctival, rectal, and vaginal washes are used to isolate the bacteria, placed in a special transport medium; the biological material must be examined no later than 24 hours after collection, as freezing and thawing can be detrimental to C. felis [50].
TREATMENT AND PROPHYLAXIS OF CHLAMYDIOSIS
Due to the systemic course of chlamydiosis, local therapy is ineffective [8]. Antibiotics such as doxycycline, azithromycin, and amoxicillin are used in long courses, supplemented by local treatment. For prophylaxis, melatonin, serotonin, or their derivatives can be used. According to some data, melatonin inhibits the initial progression of the chlamydial developmental cycle, preventing the occurrence of intracellular infection and the transformation of EBs into RBs [57]. Chlamydial bacteriophages can also be used as an alternative to antibiotics, thereby addressing the problem of antibiotic resistance. In particular, antibiotics are unable to suppress chlamydial EBs, which are metabolically inert and difficult to eradicate [42]. Chlamydiaphages cause inhibition of the developmental cycle and delay the transition of RBs to EBs, thereby reducing the possibility of infecting other cells. Chlamydial bacteriophages are found in only six species, including C. felis [43].
Vaccination for the prevention of chlamydial infection usually begins at 8–9 weeks of age, followed by revaccination after 2–4 weeks and then annually. It should be noted that information on the duration of immunity is limited. Live attenuated and inactivated adjuvanted vaccines are available on the market. There are no reliable data to compare their effectiveness. Vaccines are effective in protecting against clinical symptoms of the disease but not against the occurrence of infection. They reduce chlamydial replication in the body and lessen the clinical manifestations of infection. According to World Small Animal Veterinary Association recommendations, vaccination against chlamydiosis caused by C. felis is non-core and is recommended in cases where there is a risk of infection (outdoor access), as part of disease control measures, in crowded conditions, shelters, and for animals participating in shows and breeding [30][50][58]. No strong and convincing correlation has been found between the level of specific antibodies and the resistance of vaccinated cats to C. felis infection [50].
Immunity against the chlamydial pathogen is weak or short-term and does not protect against reinfection. Cats develop resistance to infection with age. Both humoral and cellular immunity are involved in response to chlamydial infection [8]. Cellular immunity is considered to play a decisive role in protecting the body against this pathogen. MOMP and POMP proteins are targets for protective immune responses of the body. Kittens are initially protected by antibodies for 9–12 weeks when receiving colostrum from recovered queens [8][30].
At the initial stage of the infectious process, polymorphonuclear lymphocytes are involved in the immune response. Polyclonal activation of B-lymphocytes is of primary importance in protection against chlamydiosis. Nevertheless, the leading role in immune defense against chlamydiosis belongs to T-helpers, which activate the phagocytic activity of macrophages [59].
One of the key virulence factors of Chlamydia is the proteasomal protein CPAF (chlamydial protease / proteasome-like activity factor), which suppresses neutrophil activation. In the presence of CPAF, the expression of the anti-apoptotic protein myeloid cell leukemia 1 (Mcl-1) is induced, which promotes the degradation of pro-apoptotic molecules such as BCL-2-like protein 11 (Bim). Thus, Chlamydia prevents apoptosis of host cells, leading to a prolonged period of persistence, i.e., replication inside host cells. CPAF degrades the major histocompatibility complex (MHC), hindering antigen presentation to T-cells. Furthermore, members of the genus Chlamydia increase the expression of PD-L1 (Programmed Death-Ligand 1) in host cells. The binding of PD-L1 to the PD-1 (programmed cell death protein 1) receptor on the surface of T-cells represents a negative signal that suppresses T-cell receptor (TCR) activation [60]. Thus, Chlamydia is a bacterium well-adapted to many of the host’s protective mechanisms, which complicates the process of its elimination and confirms the need for developing effective prevention methods.
CONCLUSION
Analysis of modern scientific data on feline chlamydiosis allows us to conclude that information on the epizootic situation does not reflect the true prevalence of this disease. C. felis is a pathogen capable of infecting not only cats but also other animals and humans.
The complex developmental cycle, the ability to overcome host immunity, and long-term persistence in the body complicate its eradication. Diagnosis of chlamydiosis is difficult due to the low stability of the pathogen outside the body; the most sensitive method is PCR. The long course of treatment, transition to a chronic course, and regular relapses reduce the quality of life of companion animals, and therapy requires high economic costs.
Prevention involves timely vaccination; however, immunity against the chlamydial pathogen is weak or short-lived and does not protect against reinfection. Moreover, cellular immunity is more important than humoral immunity. Hence, the need for preventive measures to protect animals from the disease arises.
For the purpose of developing successful treatment and prevention regimens for feline chlamydiosis, a more detailed study of C. felis is required.
Contribution of the authors: Tsyganov I. S. – review conceptualization, investigation, original draft writing; Shcherbinin S. V. – investigation, original draft writing, draft editing; Galkina Т. S. – supervision, draft editing; Gruzdev K. N. – supervision, draft editing.
Вклад авторов: Цыганов И. С. – концепция обзора, проведение поисково-аналитической работы, подготовка текста статьи; Щербинин С. В. – проведение поисково-аналитической работы, подготовка текста статьи, редактирование статьи; Галкина Т. С. – научное руководство, редактирование статьи; Груздев К. Н. – научное консультирование, редактирование текста статьи.
References
1. Sostaric-Z uckermann I. C., Borel N., Kaiser C., Grabarevic Z., Pospischil A. Chlamydia in canine or feline coronary arteriosclerotic lesions. BMC Research Notes. 2011; 4:350. https://doi.org/10.1186/1756-0500-4-350
2. Sibitz C., Rudnay E. C., Wabnegger L., Spergser J., Apfalter P., Nell B. Detection of Chlamydophila pneumoniae in cats with conjunctivitis. Veterinary Ophthalmology. 2011; 14 (Suppl. 1): 67–74. https://doi.org/10.1111/j.1463-5224.2011.00919.x
3. Fukushi H., Hirai K. Genetic diversity of avian and mammalian Chlamydia psittaci strains and relation to host origin. Journal of Bacteriology. 1989; 171 (5): 2850–2855. https://doi.org/10.1128/jb.171.5.2850-2855.1989
4. Sanderson H., Vasquez M., Killion H., Vance M., Sondgeroth K., Fox J. Fatal Chlamydia psittaci infection in a domestic kitten. Journal of Veterinary Diagnostic Investigation. 2021; 33 (1): 101–103. https://doi.org/10.1177/1040638720966960
5. Lewin A. C., Hicks S. K., Carter R. T. A review of evidence- based management of infectious ocular surface disease in shelter- housed domestic cats. Veterinary Ophthalmology. 2023; 26 (Suppl. 1): 47–58. https://doi.org/10.1111/vop.13063
6. Chan I., Dowsey A., Lait P., Tasker S., Blackwell E., Helps C. R., Barker E. N. Prevalence and risk factors for common respiratory pathogens within a cohort of pet cats in the UK. Journal of Small Animal Practice. 2023; 64 (9): 552–560. https://doi.org/10.1111/jsap.13623
7. Longbottom D., Coulter L. J. Animal chlamydioses and zoonotic implications. Journal of Comparative Pathology. 2003; 128 (4): 217–244. https://doi.org/10.1053/jcpa.2002.0629
8. Sykes J. E. Feline chlamydiosis. Clinical Techniques in Small Animal Practice. 2005; 20 (2): 129–134. https://doi.org/10.1053/j.ctsap.2004.12.018
9. Halánová M., Sulinová Z., Čisláková L., Trbolová A., Páleník L., Weissová T., et al. Chlamydophila felis in cats – are the stray cats dangerous source of infection? Zoonoses and Public Health. 2011; 58 (7): 519–522. https://doi.org/10.1111/j.1863-2378.2011.01397.x
10. Fontbonne A. Infertility in queens: Clinical approach, experiences and challenges. Journal of Feline Medicine and Surgery. 2022; 24 (9): 825–836. https://doi.org/10.1177/1098612x221118752
11. Sachse K., Bavoil P. M., Kaltenboeck B., Stephens R. S., Kuo C.-C., Rosselló- Móra R., Horn M. Emendation of the family Chlamydiaceae: proposal of a single genus, Chlamydia, to include all currently recognized species. Systematic and Applied Microbiology. 2015; 38 (2): 99–103. https://doi.org/10.1016/j.syapm.2014.12.004
12. Wu S.-M., Huang S.-Y., Xu M.-J., Zhou D.-H., Song H.-Q., Zhu X.-Q. Chlamydia felis exposure in companion dogs and cats in Lanzhou, China: A public health concern. BMC Veterinary Research. 2013; 9:104. https://doi.org/10.1186/1746-6148-9-104
13. Ulbert Á. B., Juhász H., Karácsony Z., Bencze K., Deim Z., Burián K., Terhes G. The occurrence of Chlamydia felis in cats and dogs in Hungary. Pathogens. 2024; 13 (9):771. https://doi.org/10.3390/pathogens13090771
14. Hughes L., Visser S., Heddema E., de Smet N., Linssen T., Wijdh R. J., Huis in ‘t Veld R. Zoonotic transmission of Chlamydia felis from domestic cats; A case series of chronic follicular conjunctivitis in humans. New Microbes and New Infections. 2024; 59:101412. https://doi.org/10.1016/j.nmni.2024.101412
15. Jazi S., Mokhtari A., Kahrizsangi A. E. Molecular detection of Chlamydia psittaci and Chlamydia felis in human keratoconjunctivitis cases. Bulgarian Journal of Veterinary Medicine. 2020; 23 (1): 130–137. https://doi.org/10.15547/bjvm.2124
16. Wons J., Meiller R., Bergua A., Bogdan C., Geißdörfer W. Follicular conjunctivitis due to Chlamydia felis – case report, review of the literature and improved molecular diagnostics. Frontiers in Medicine. 2017; 4:105. https://doi.org/10.3389/fmed.2017.00105
17. Chen J., Long J., Zhou H., Huang C., Zhu Y., Wang R., et al. Isolation and characterization of Chlamydia felis and its pathogenesis in cats. Veterinary Microbiology. 2024; 295:110128. https://doi.org/10.1016/j.vetmic.2024.110128
18. Miyashita N., Fukano H., Mouri K., Fukuda M., Yoshida K., Kobashi Y., et al. Community- acquired pneumonia in Japan: a prospective ambulatory and hospitalized patient study. Journal of Medical Microbiology. 2005; 54 (4): 395–400. https://doi.org/10.1099/jmm.0.45920-0
19. Corsaro D., Venditti D. Detection of novel Chlamydiae and Legionellales from human nasal samples of healthy volunteers. Folia Microbiologica. 2015; 60 (4): 325–334. https://doi.org/10.1007/s12223-015-0378-y
20. Laroucau K., Di Francesco A., Vorimore F., Thierry S., Pingret J. L., Bertin C., et al. Multilocus variable- number tandem- repeat analysis scheme for Chlamydia felis genotyping: comparison with multilocus sequence typing. Journal of Clinical Microbiology. 2012; 50 (6): 1860–1866. https://doi.org/10.1128/jcm.00417-12
21. Marti I., Pisano S. R. R., Wehrle M., Meli M. L., Hofmann- Lehmann R., Ryser-D egiorgis M. P. Severe conjunctivitis associated with Chlamydia felis infection in a free-ranging Eurasian lynx (Lynx lynx). Journal of Wildlife Diseases. 2019; 55 (2): 522–525. https://doi.org/10.7589/2018-05-142
22. Luu L. D. W., Kasimov V., Phillips S., Myers G. S. A., Jelocnik M. Genome organization and genomics in Chlamydia: whole genome sequencing increases understanding of chlamydial virulence, evolution, and phylogeny. Frontiers in Cellular and Infection Microbiology. 2023; 13:1178736. https://doi.org/10.3389/fcimb.2023.1178736
23. Feodorova V. A. Genomnye i postgenomnye tekhnologii v izuchenii i diagnostike khlamidiozov = Genomic and postgenomic technologies in the study and diagnosis of chlamydial infections. Metody komp’yuternoi diagnostiki v biologii i meditsine – 2020: sbornik statei Vserossiiskoi shkoly-seminara (Saratov, 18–19 noyabrya 2020 g.) = Methods of computer diagnostics in biology and medicine – 2020: proceedings of the All- Russian school-s eminar (Saratov, November 18–19, 2020). Saratov: Saratovskii istochnik; 2020; 19–22. https://elibrary.ru/jgjgai (in Russ.)
24. McManus C. M., Levy J. K., Andersen L. A., McGorray S. P., Leutenegger C. M., Gray L. K., et al. Prevalence of upper respiratory pathogens in four management models for unowned cats in the Southeast United States. The Veterinary Journal. 2014; 201 (2): 196–201. https://doi.org/10.1016/j.tvjl.2014.05.015
25. Ju H., Yang D., Jin J., Wang J., Li X., Yang X., et al. Spectrum detection and analysis of the epidemiological characteristics of infectious pathogens in the feline respiratory tract. Archives of Virology. 2024; 169 (9):177. https://doi.org/10.1007/s00705-024-06093-5
26. Ohya K., Takahara Y., Kuroda E., Koyasu S., Hagiwara S., Sakamoto M., et al. Chlamydophila felis CF0218 is a novel TMH family protein with potential as a diagnostic antigen for diagnosis of C. felis infection. Clinical and Vaccine Immunology. 2008; 15 (10): 1606–1615. https://doi.org/10.1128/cvi.00134-08
27. Kiełbowicz Z., Płoneczka- Janeczko K., Bania J., Bierowiec K., Kiełbowicz M. Characteristics of the bacterial flora in the conjunctival sac of cats from Poland. Journal of Small Animal Practice. 2015; 56 (3): 203–206. https://doi.org/10.1111/jsap.12304
28. Konyaev S. V. Prevalence of causative agents of respiratory infections in cats and dogs in Russia. Russian Veterinary Journal. 2020; (1): 9–13. https://doi.org/10.32416/2500-4379-2020-20201-9-13 (in Russ.)
29. Strugovschikov A. Yu., Pudovkin N. A., Salautin V. V. Features of the spread of Chlamydia infection in Moscow. International Bulletin of Veterinary Medicine. 2020; (2): 21–25. https://elibrary.ru/qikmft (in Russ.)
30. Gruffydd- Jones T., Addie D., Belák S., Boucraut-B aralon C., Egberink H., Frymus T., et al. Chlamydophila felis infection: ABCD guidelines on prevention and management. Journal of Feline Medicine and Surgery. 2009; 11 (7): 605–609. https://doi.org/10.1016/j.jfms.2009.05.009
31. Azuma Y., Hirakawa H., Yamashita A., Cai Y., Rahman M. A., Suzuki H., et al. Genome sequence of the cat pathogen, Chlamydophila felis. DNA Research. 2006; 13 (1): 15–23. https://doi.org/10.1093/dnares/dsi027
32. Feodorova V. A., Lyapina А. М., Khizhnyakova М. А., Zaitsev S. S., Saltykov Y. V., Subbotina I. А., et al. Chlamydia of animals and humans. Moscow: Nauka; 2019. 135 p. https://doi.org/10.7868/9785020402492 (in Russ.)
33. Borel N., Polkinghorne A., Pospischil A. A review on chlamydial diseases in animals: still a challenge for pathologists? Veterinary Pathology. 2018; 55 (3): 374–390. https://doi.org/10.1177/0300985817751218
34. Ravichandran K., Anbazhagan S., Karthik K., Angappan M., Dhayananth B. A comprehensive review on avian chlamydiosis: a neglected zoonotic disease. Tropical Animal Health and Production. 2021; 53 (4):414. https://doi.org/10.1007/s11250-021-02859-0
35. Bressan M., Rampazzo A., Kuratli J., Marti H., Pesch T., Borel N. Occurrence of Chlamydiaceae and Chlamydia felis pmp9 typing in conjunctival and rectal samples of Swiss stray and pet cats. Pathogens. 2021; 10 (8):951. https://doi.org/10.3390/pathogens10080951
36. Vafin R. R., Ravilov R. H., Gaffarov H. Z., Ravilov A. Z., Iskhakov G. M., Bakirov I. H. Comparative characteristic of the strains of khlamidiosis on the omp1-gene. Veterinarnaya praktika. 2007; (3): 54–59. https://elibrary.ru/knpdej (in Russ.)
37. Harley R., Herring A., Egan K., Howard P., Gruffydd- Jones T., Azuma Y., et al. Molecular characterisation of 12 Chlamydophila felis polymorphic membrane protein genes. Veterinary Microbiology. 2007; 124 (3–4): 230–238. https://doi.org/10.1016/j.vetmic.2007.04.022
38. Di Francesco A., Baldelli R. Feline chlamydiosis in Italy: PCR amplification and analysis of the ompA and groEL-homolog genes. New Microbiologica. 2002; 25 (3): 341–344. https://pubmed.ncbi.nlm.nih.gov/12173777
39. Semenov V. M., Semenov D. M., Khvorik D. F., Kozin V. N., Bazhin Yu. A., Dmitrachenko T. I., et al. Chlamydia infection. Vitebsk: Vitebsk State Order of Peoples’ Friendship Medical University; 2006. 205 p. https://elibrary.ru/emcwop (in Russ.)
40. Aldyakov A. V., Konanova T. E. Chlamidiosis in cats. Vestnik Chuvash State Agricultural Academy. 2021; (2): 53–57. https://elibrary.ru/nsylpm (in Russ.)
41. Ravilov A. Z., Ghaffarov Kh. Z., Ravilov R. H. Chlamydiosis in animals. Kazan: Fen; 2004. 368 p. (in Russ.)
42. Klose S. M., De Souza D. P., Devlin J. M., Bushell R., Browning G. F., Vaz P. K. A “plus one” strategy impacts replication of felid alphaherpesvirus 1, Mycoplasma and Chlamydia, and the metabolism of coinfected feline cells. mSystems. 2024; 9 (10): e00852-24. https://doi.org/10.1128/msystems.00852-24
43. Pawlikowska- Warych M., Śliwa-Dominiak J., Deptuła W. Chlamydial plasmids and bacteriophages. Acta Biochimica Polonica. 2015; 62 (1): 1–6. https://doi.org/10.18388/abp.2014_764
44. Gladin D. P., Korolyuk A. M., Drobot I. V., Kirillova N. P., Kozlova N. S., Annenkova I. D. Chlamydia and chlamydiosis. Russian Biomedical Research. 2021; 6 (4): 37–46. https://elibrary.ru/olcqbo (in Russ.)
45. Elwell C., Mirrashidi K., Engel J. Chlamydia cell biology and pathogenesis. Nature Reviews Microbiology. 2016; 14 (6): 385–400. https://doi.org/10.1038/nrmicro.2016.30
46. Cheong H. C., Lee C. Y. Q., Cheok Y. Y., Tan G. M. Y., Looi C. Y., Wong W. F. Chlamydiaceae: diseases in primary hosts and zoonosis. Microorganisms. 2019; 7 (5):146. https://doi.org/10.3390/microorganisms7050146
47. Lisova V., Savchenko A. Pathomorphological characteristics of chlamydiosis in cats. Scientific Messenger LNUVMBT named after S. Z. Gzhytskyj. 2017; 19 (77): 11–14. https://elibrary.ru/ytalpz (in Ukrainian)
48. Caspe S. G., Hill H. Chlamydiosis in animals. Animals. 2024; 14 (21):3130. https://doi.org/10.3390/ani14213130
49. Nguyen D., Barrs V. R., Kelman M., Ward M. P. Feline upper respiratory tract infection and disease in Australia. Journal of Feline Medicine and Surgery. 2019; 21 (10): 973–978. https://doi.org/10.1177/1098612x18813248
50. Diagnosis and prevention of infectious diseases in dogs and cats: a guide for veterinary practitioners. Ed. by T. I. Aliper. Moscow: Zoovetkniga; 2017. 300 p. (in Russ.)
51. Wasissa M., Lestari F. B., Nururrozi A., Tjahajati I., Indarjulianto S., Salasia S. I. O. Investigation of chlamydophilosis from naturally infected cats. Journal of Veterinary Science. 2021; 22 (6): e67. https://doi.org/10.4142/jvs.2021.22.e67
52. Lisova V., Savchenko A. Histological changes in cats at chlamydiosis. Scientific Messenger LNUVMB. 2017; 19 (78): 158–161. https://elibrary.ru/zswrst (in Ukrainian)
53. Mills D. Diagnosis of Chlamydophila felis by conjunctival cytology in shelter cats. BSAVA Congress Proceedings. 2016; 527. https://www.bsavalibrary.com/content/chapter/10.22233/9781910443446.ch66sec5
54. Belova E. V., Kapustina T. A., Markina A. N., Parilova O. V. Laboratory diagnostics of respiratory chlamydia. Siberian Medical Review. 2019; (1): 5–16. https://doi.org/10.20333/2500136-2019-1-5-16 (in Russ.)
55. Scidmore M. A. Cultivation and laboratory maintenance of Chlamydia trachomatis. Current Protocols in Microbiology. 2006; 11A.1.1–11A.1.25. https://doi.org/10.1002/9780471729259.mc11a01s00
56. Zur N. V., Mironov A. Yu., Aleshkin V. A., Afanasjev S. S., Rubalskaya E. E., Afanasjev M. S., Rubalskii E. O. Actual aspects of laboratory diagnostics of urogenital chlamydial infections. Astrakhan Medical Journal. 2016; 11 (2): 16–32. https://elibrary.ru/wfaqab (in Russ.)
57. Rahman M. A., Azuma Y., Fukunaga H., Murakami T., Sugi K., Fukushi H., et al. Serotonin and melatonin, neurohormones for homeostasis, as novel inhibitors of infections by the intracellular parasite Chlamydia. Journal of Antimicrobial Chemotherapy. 2005; 56 (5): 861–868. https://doi.org/10.1093/jac/dki331
58. Squires R. A., Crawford C., Marcondes M., Whitley N. 2024 guidelines for the vaccination of dogs and cats – compiled by the Vaccination Guidelines Group (VGG) of the World Small Animal Veterinary Association (WSAVA). Journal of Small Animal Practice. 2024; 65 (5): 277–316. https://doi.org/10.1111/jsap.13718
59. Medova E. V., Pivovarenko E. A. Razreshayushchaya sposobnost’ metodov prizhiznennoi diagnostiki khlamidiinoi infektsii v populyatsii plotoyadnykh v usloviyakh urbanizirovannykh territorii = Sensitivity of ante-mortem diagnostic methods for chlamydial infection in carnivore populations within urban environments. Russian Journal of Veterinary Pathology. 2005; (4): 132–134. https://elibrary.ru/hsqenl (in Russ.)
60. Wong W. F., Chambers J. P., Gupta R., Arulanandam B. P. Chlamydia and its many ways of escaping the host immune system. Journal of Pathogens. 2019; 2019:8604958. https://doi.org/10.1155/2019/8604958
About the Authors
Ilya S. TsyganovRussian Federation
Ilya S. Tsyganov, Postgraduate Student, Veterinarian, Laboratory for Pets Diseases Prevention,
ul. Gvardeyskaya, 6, Yur’evets, Vladimir 600901.
Sergey V. Shcherbinin
Russian Federation
Sergey V. Shcherbinin, Cand. Sci. (Veterinary Medicine), Researcher, Information and Analysis Centre,
ul. Gvardeyskaya, 6, Yur’evets, Vladimir 600901.
Tatyana S. Galkina
Russian Federation
Tatyana S. Galkina, Cand. Sci. (Veterinary Medicine), Head of Laboratory for Pets Diseases Prevention,
ul. Gvardeyskaya, 6, Yur’evets, Vladimir 600901.
Konstantin N. Gruzdev
Russian Federation
Konstantin N. Gruzdev, Dr. Sci. (Biology), Professor, Chief Researcher, Information and Analysis Centre,
ul. Gvardeyskaya, 6, Yur’evets, Vladimir 600901.
Review
For citations:
Tsyganov I.S., Shcherbinin S.V., Galkina T.S., Gruzdev K.N. Feline chlamydiosis (review). Veterinary Science Today. 2025;14(4):326-336. https://doi.org/10.29326/2304-196X-2025-14-4-326-336
JATS XML



































